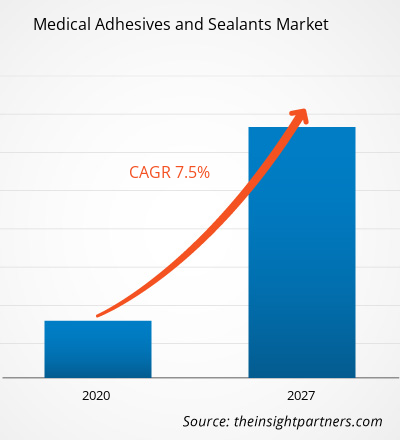
Medical-grade Silicone Market is witnessing a steady transformation driven by the modernization of healthcare systems and the rise of minimally invasive surgeries as of March 12, 2026. Valued at USD 0.34 billion in 2024, the industry is projected to grow from USD 0.3566 billion in 2025 to USD 0.5738 billion by 2035. This represents a consistent compound annual growth rate (CAGR) of 4.8% during the forecast period.
Medical-grade silicones are prized for their biocompatibility, chemical inertness, and ability to withstand repeated sterilization. In 2026, the market is being catalyzed by the “Personalized Healthcare” trend, where silicones are critical for customized orthotics, high-precision prosthetic liners, and wearable diagnostic sensors that require prolonged skin contact.
GLOBAL SUPPLY CHAIN & MARKET DISRUPTION ALERT
Escalating geopolitical tensions in the Middle East, particularly around the Strait of Hormuz and the Red Sea, are creating significant disruptions across global energy, chemicals, and logistics markets. Critical shipping corridors are under pressure, with major oil, LNG, petrochemical, and raw material flows at risk, triggering supply chain delays, freight cost surges, insurance withdrawals, and heightened price volatility. These disruptions are increasing operational risks and cost uncertainties for industries dependent on global trade routes and energy-linked feedstocks.
Access our real-time disruption analysis covering supply chain risks, price outlook scenarios, logistics impacts, and alternative sourcing strategies.
👉 Request a Sample Report to understand the potential impact on your industry and get actionable insights to support procurement, risk management, and supply chain planning.
Key Market Trends & Insights (2026 Update)
-
Resilient Valuation Growth: Data indicates the market scaling toward USD 0.5738 billion by 2035. The 4.8% CAGR is firmly anchored by the Medical Devices and Orthopedics sectors, which prioritize high-purity elastomers and gels.
-
The Wearable Sensor Boom: In early 2026, demand for Medical Adhesives and Coatings has surged. This is driven by the expansion of the remote patient monitoring market, where skin-friendly silicone adhesives are required for continuous glucose monitors (CGM) and cardiac patches.
-
Advancements in Contact Lenses: 2026 industry insights highlight a shift toward silicone hydrogel lenses with enhanced oxygen permeability. This segment remains a significant consumer of high-grade silicone monomers to ensure long-term eye health and comfort.
-
Precursor and Logistics Pressures: As of March 12, 2026, the cost of High-Purity Polysiloxanes has risen by 10–13% due to the logistical disruptions noted in our Global Disruption Alert. This is leading to localized pricing adjustments for medical-grade tubing and components.
-
Orthotics & Prosthetics Customization: In 2026, Silicone Gels are seeing record adoption in the prosthetics industry. Their ability to distribute pressure and reduce shear force is essential for the comfort of next-generation prosthetic limbs.
Get Sample Report PDF:https://www.marketresearchfuture.com/sample_request/5767
Market Segmentation
-
By Product:
-
Elastomers: The largest segment, used for tubing, catheters, seals, and implants.
-
Gels: Critical for prosthetic liners, scar management, and cushioning.
-
Medical Adhesives: Essential for wound care and wearable medical patches.
-
Medical Coatings: Used to enhance the lubricity and biocompatibility of surgical tools.
-
-
By Application:
-
Medical Devices: Catheters, valves, respiratory masks, and drug delivery systems.
-
Medical Tapes: Skin-safe tapes for sensitive and neonatal care.
-
Orthopedics & Prosthetics: Soft-tissue replacement and comfort liners.
-
Contact Lenses: High-permeability silicone hydrogel materials.
-
-
By Region:
-
North America: Leading the market with advanced R&D in implantable silicones and aesthetic medicine.
-
Asia-Pacific: The fastest-growing region in 2026, fueled by expanding healthcare infrastructure in China and India.
-
Europe: Focused on strict regulatory compliance (MDR) and high-performance surgical silicones.
-
Strategic Industry Analysis (2026 Context)
In 2026, the medical-grade silicone sector is defined by “Regulatory Precision.” With the full implementation of updated 2026 global health standards, manufacturers are prioritizing “Clean-Room Certified” silicones that guarantee zero extractables or leachables. As mentioned in our Global Disruption Alert, the volatility of global shipping has favored Regional Medical-Grade Compounding. In March 2026, we are seeing a shift where medical device OEMs are qualifying localized silicon suppliers to avoid the freight surcharges and transit delays currently affecting intercontinental shipping.
Technically, 2026 is seeing the rollout of “Antimicrobial Silicones.” These advanced elastomers incorporate silver-ion or other bioactive technologies directly into the silicone matrix, significantly reducing the risk of device-related infections in long-term catheters and wound drainage systems.
Related Reports
People Also Ask: FAQ
What is the medical-grade silicone market size in 2026?
The market is currently valued at approximately USD 0.37 billion in 2026, growing from its USD 0.34 billion base in 2024. It is projected to reach USD 0.5738 billion by 2035.
What is the CAGR for medical-grade silicone?
The industry is exhibiting a steady CAGR of 4.8% during the forecast period of 2025–2035, driven by the aging global population and the rise of wearable medical tech.
How do Red Sea disruptions affect medical silicone prices in 2026?
As of March 2026, increased freight and insurance costs for high-purity chemical transport have led to a 10–12% price hike for medical-grade resins in markets that rely on intercontinental shipping.
Is medical-grade silicone safe for long-term implants in 2026?
Yes. In 2026, specific Long-Term Implantable (LTI) grades are rigorously tested and certified for biocompatibility, ensuring they do not cause adverse reactions when remaining in the body for extended periods.
Why is silicone preferred for wearable medical patches?
In 2026, silicone adhesives are the gold standard for wearables because they provide a secure bond while being “atraumatic”—meaning they can be removed from sensitive skin without causing irritation or damage.